Coincidentally, some model engineering forums I follow have been showing examples of bronze valve cages 'glued' into aluminum heads with various adhesives. Before you load the verbal assault rifles (LOL) some background info. We actually know what we are doing! Well... kind of. These are fussy little buggers. The typical 45-deg valve seat is only 0.010-0.015". So pressing them into the heads with any kind of interference is usually a recipe for distortion, ovaling = valve leak, maybe a junked head - a real headache. Yes the seats might be re-cut & re-lapped, but its much more desirable to establish the seal beforehand, then insert the cage stress free retained with a thin annular film of adhesive.
Where was I? Oh ya, JB weld. Some folks have subjected coupons to fire & fury tests with a blowtorch & wrench, simulating combustion chamber conditions & exhaust port couplings. Apparently JB is standing up better than most of the high temp Loctite's & HT retaining compounds, observing all the gap widths & cleanliness. This was interesting to me, so I intend to repeat. The metal filler is probably helping, but I've had some experience with true high temp resins & they inevitably have to be post cured. So story to be continued.
One thing I can pass on from my RC model days & probably have mixed 10,000 epoxy batches. The smaller the quantity, the harder it is to get the correct 1:1 resin/hardener mix. They are a little bit tolerant of misproportion, but not much. I know when it comes to laminating epoxies like composite structures, even 5% misproportion can cause problems, improper cure or not attaining full strength. So image 5% of a 2 dots of JB. What I find helps is to squeeze them out in parallel lines beside one another so you can eyeball volume better. I use a clean sheet of notepad paper, no used materials especially if they are dirty or greasy.
Another thing is to very thoroughly mix the 2 components. Don't just give it a few swirls & call it good. Don't run yourself out of application time of course, but I would mix for at least 30 secs to a minute, again with a clean stir stick. Some folks have tried gentle heat & post cure heat, but I haven't seen any tangible specs (on JB). For sure this is a known thing in laminating resins.
For six years with a medical device company, I lived and breathed epoxy.
Chemically, an epoxy is a compound formed by the reaction between an epoxide (the resin) and an amine (the hardener) The structure of the hardener determines what the mechanical properties of the final product are. Typically, the hardener has three reaction sites which make for a highly cross linked product. In addition to the resin and hardener, additives are used to create the final properties.
One fact that is little known is that an epoxy that is deemed "cured" actually has only completed about 10% of the chemical reaction. When the epoxy is first mixed, the molecules are free to move about and re-orientate into a "docking" position. As the reaction continues, the number of available sites decreases and it becomes progressively harder for the remaining reactants to react. Typically, a completed bond must break and new bonds must occur for the reaction to continue. For the resins which we used, the "cure" time was the order of several hours but the time required for a 90% cure stretched into years. A newly "cured" resin can actually be liquified by raising its temperature. As the epoxy continues to cure, the temperature at which liquification occurs rises. A common practice for us was to cure as close to room temperature as possible until the epoxy set and then cure overnight at 105ºF. A "final" cure was to elevate the temperature to 230ºF for several days. At that point, we were probably at about 60% of a complete reaction.
As with most chemical reactions, raising the temperature decreases the cure time. As a rule of thumb, 10ºC rise in temperature will double the reaction rate. Add to this that the chemical reaction gives off heat which can raise the internal temperature significantly for larger mix volumes.
So what does this mean for the casual user?
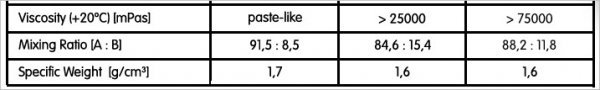
First, from our observations, a 5% variation in composition will have no significant effect on final product mechanical properties. For one product, we purposely changed the mixes by 3, 6, and 10%. Improper mixing and incomplete cure has a more profound effect. Nevertheless, when I mix small amounts of epoxy for critical applications, I will weigh them on an analytical balance capable of weighing to .1 milligrams. While the densities of the two components are usually close, for critical work, it would be prudent to determine the actual densities as most epoxy recipes are by volume.
Second, thorough mixing is important to avoid pockets of unmixed reactants, I will usually use at least a gram of each to ensure having enough bulk to properly mix. I mix until I think it is done and then do it again. The technique that I use is to mix on a piece of plastic sheet (coffee can lids work well) mixing the components with a spatula made from a clean tongue depressor with ine end cut flat. I then spread the mixture to a thin layer and scrape it into a lump for a second mixing. I repeat this several times. Using this technique, I don't end up with uncured epoxy on the plastic sheet which is a good indication that the epoxy that I used was well mixed. I always leave a small amount for a test sample to check for curing.
Third, wherever possible, allow the epoxy to cure at room temperature until hard and the cure at an elevated temperature. I would slowly ramp up my temperatures to 230ºF. This process improves the high temperature characteristics of the epoxy and it gives a harder epoxy at room temperature.
Finally, a variety of additives are used to enhance various mechanical properties, iron dust, aluminum dust, and glass microspheres to name a few. Talc and carbon black are used to control uncured flow properties and as a visual guide to proper mixing. I have used wood flour mixed with the epoxy to patch defects in wood items. They will impact the mixing strategy, particularly if the components are mixed by weight rather than volume.